Ever wondered what is green hydrogen and what is all the fuss around it? As a recent member of the Women in Green Hydrogen community, let us take a deep dive on how clean hydrogen is made and later on look at the role geosscience has to play.
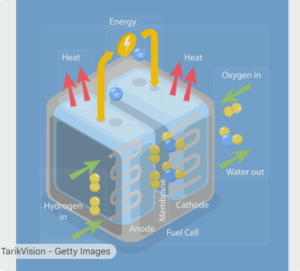
This diagram illustrates the operation of a fuel cell, a device that electrochemically converts chemical energy from a fuel, such as hydrogen, into electricity. The process involves two electrodes, an anode and a cathode, separated by a membrane or electrolyte. The fuel cell operates without combustion, making it a clean and efficient energy source.
The Process
The fuel cell works by separating the fuel and the oxidant and facilitating their reaction in a controlled manner. Hydrogen is supplied to one electrode, and oxygen to the other. The overall reaction produces water, heat, and electrical energy. The components labeled are the anode, cathode, membrane, and fuel cell.
Hydrogen Fuel In
The process begins with hydrogen gas being introduced into the fuel cell at the anode. At this point, the hydrogen molecules are split into protons (hydrogen ions) and electrons. This separation is a critical first step in the energy conversion process.
The Membrane’s Role
A special membrane separates the anode and cathode, allowing only the positively charged hydrogen ions (protons) to pass through. The electrons are blocked by the membrane and are forced to travel through an external circuit to the cathode. This movement of electrons constitutes the electric current.
Generating Electrical Energy
As the electrons travel through the external circuit, they create an electrical current, which can be used to power devices. This flow of electrons from the anode to the cathode is what generates the useful electrical energy produced by the fuel cell.
Oxygen In and Water Out
Simultaneously, oxygen is supplied to the cathode side of the fuel cell. At the cathode, the oxygen molecules react with the protons that have passed through the membrane and the electrons that have traveled through the external circuit. This reaction forms water, which is the primary byproduct of the fuel cell.
Heat as a Byproduct
In addition to electricity and water, the electrochemical reaction within the fuel cell also generates heat. This heat is a byproduct of the energy conversion process. In some applications, this waste heat can be captured and used for heating, a process known as cogeneration or combined heat and power (CHP).
Hope that was a helpful introduction to green hydrogen


